Amino Acid: Understanding Hydrophobic And Hydrophilic Properties
Amino acids are the building blocks of proteins, and their unique properties play a crucial role in biological functions. Among these properties, the hydrophobic and hydrophilic characteristics of amino acids significantly influence how proteins fold, interact, and function within living organisms. Each amino acid possesses a side chain, or R group, that determines whether it is hydrophobic, hydrophilic, or has amphipathic properties. Understanding these distinctions is vital for biochemists and molecular biologists as they explore the complexities of protein structures and their interactions in various environments.
In this article, we will delve into the fascinating world of amino acids, exploring the differences between hydrophobic and hydrophilic amino acids, their significance in protein structure, and how they contribute to various biological processes. By understanding these concepts, we can appreciate the intricate dance of molecules that sustains life and recognizes how these interactions can influence everything from enzyme activity to signal transduction pathways.
Join us as we answer essential questions about amino acids, such as: What makes an amino acid hydrophobic or hydrophilic? How do these properties affect protein structure? And why is it crucial for scientists to comprehend these characteristics? Let’s get started on this biochemical journey!
What are Hydrophobic and Hydrophilic Amino Acids?
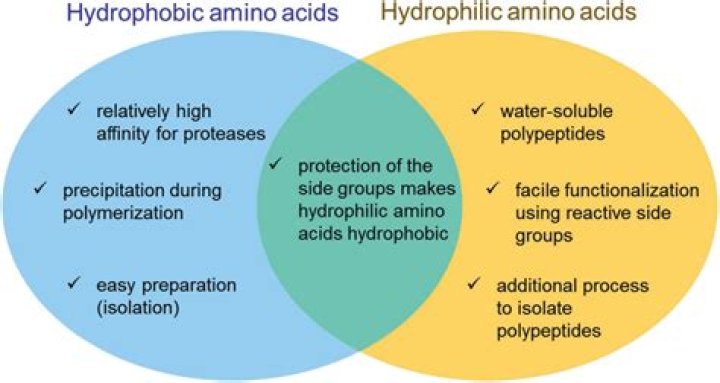
Hydrophobic and hydrophilic amino acids are classified based on the properties of their side chains. Hydrophobic amino acids have non-polar side chains that do not interact favorably with water, while hydrophilic amino acids possess polar or charged side chains that readily interact with water. This distinction is essential for understanding how proteins fold and function within an aqueous environment.
Why Are Amino Acids Classified as Hydrophobic or Hydrophilic?
The classification of amino acids as hydrophobic or hydrophilic is primarily based on their side chain properties. The side chains can vary significantly in structure and charge, influencing how they behave in aqueous solutions. This classification affects protein folding, stability, and functionality, making it a crucial aspect of biochemistry.
What Role Do Hydrophobic Amino Acids Play in Protein Structure?
Hydrophobic amino acids tend to cluster together in the interior of protein structures, away from the aqueous environment. This clustering stabilizes the protein's three-dimensional shape, which is vital for its biological function. The interactions between hydrophobic side chains help maintain the overall structure of proteins, contributing to their stability and functionality.
How Do Hydrophilic Amino Acids Contribute to Protein Function?
Hydrophilic amino acids, with their polar and charged side chains, often reside on the surface of proteins, interacting with the aqueous environment and other polar molecules. These interactions can facilitate enzyme-substrate binding, receptor-ligand interactions, and other critical biological processes. Understanding the role of hydrophilic amino acids is essential for grasping how proteins carry out their functions in the body.
What Are Some Examples of Hydrophobic and Hydrophilic Amino Acids?
Here are some common examples of hydrophobic and hydrophilic amino acids:
- Hydrophobic Amino Acids:
- Alanine (Ala)
- Leucine (Leu)
- Isoleucine (Ile)
- Phenylalanine (Phe)
- Valine (Val)
- Hydrophilic Amino Acids:
- Serine (Ser)
- Threonine (Thr)
- Glutamine (Gln)
- Lysine (Lys)
- Aspartic acid (Asp)
How Can Understanding Amino Acid Properties Aid in Drug Design?
Understanding the hydrophobic and hydrophilic properties of amino acids can significantly enhance drug design efforts. By analyzing the interactions between drugs and target proteins, researchers can design more effective therapeutic agents that fit snugly into their target sites, improving efficacy and reducing side effects. Knowledge of these properties can lead to the development of drugs that better mimic natural substrates or inhibitors.
Conclusion: The Importance of Amino Acid Properties in Biochemistry
In summary, the classification of amino acids as hydrophobic or hydrophilic is fundamental to understanding protein structure and function. These properties influence how proteins fold, interact with other molecules, and carry out their roles within living organisms. As we continue to explore the complex world of biochemistry, the significance of amino acids—both hydrophobic and hydrophilic—will remain a critical area of study, with implications for everything from basic biological research to drug design and therapeutic advancements.
Unveiling The Magic Of Pinoy Flix Teleserye
Unveiling The Mystery: What Is Ebube Obio Age?
Exploring MrBeast's Relationships: The Man Behind The Philanthropy
TJ. Hydrophobic Amino Acids. Amino acids are grouped according to what
Do hydrophobic amino acids have nonpolar side chains solelasem
Pin on Amino Acids


